The European Commission recently published the pharmaceutical legislative package (a proposal of Directive repealing Directive 2001/83/EC and Directive 2009/35/EC (Directive proposal) and a proposal of Regulation amending Regulations (EC) 1394/2007 and (EU) 536/2014, and repealing Regulations (EC) 726/2004, (EC) 141/2000 and (EC) 1901/2006) (Regulation proposal)) which could be the largest reform in over 20 years in this area.
The proposal induces, in particular, major changes in data protection and market exclusivity suggesting the willingness to reinforce the conception of these periods as rewards and not as a given. The data protection provisions are mainly included in the Directive proposal although the Regulation proposal contains some provisions on this matter in particular regarding the specific cases of orphan and paediatric medicinal products.
In this context, the Commission proposes to reduce the baseline data protection period while extending the number of possibilities of prolongation of this period. If the concerns raised by the pharmaceutical industry are understandable, it should be noted that the additional prolongation periods for data protection relate to subjects of major interest to the patients. In particular, improved access, which is fundamentally the objective of the reform guiding these proposals. In the future, 6 years after the granting of a medicinal product's marketing authorisation ("MA"), the data protection attached to such product will have to be earned.
This article details below the content of the proposals regarding data protection and market exclusivity (1.) before considering particular cases (2.).
1. Data protection and market exclusivity according the proposal of the Commission
The Directive proposal maintains the concept of a global marketing authorisation (Directive proposal, Article 5) - knowing that the new temporary emergency MA (i.e. the MA granted to a product with a view to addressing public health emergencies) and any subsequent MA, shall also be considered as part of the same global MA (Regulation proposal, Article 36). Such proposal would substantially affect the baseline period (1.1), but would however allow prolongations subject to conditions (1.2)1. These new principles are intended to apply to both national and centralized authorised products, subject to exceptions.
1.1 Reduction of the baseline period for data protection
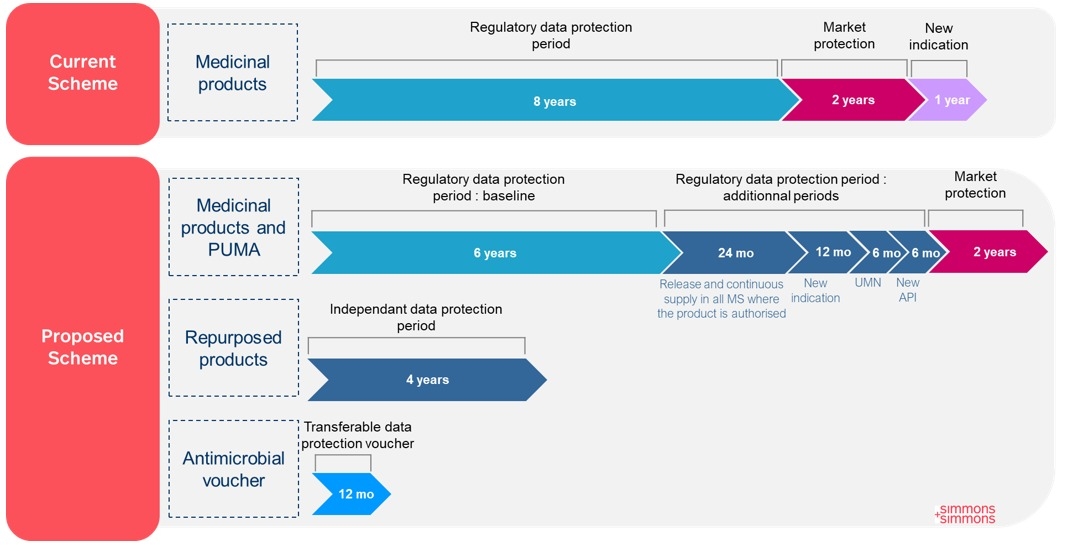
The regulatory data protection corresponds to the period during which another applicant cannot refer to the data of an original MA to submit a new MA.
The Commission proposes to reduce the baseline period of regulatory data protection from 8 years to 6 years (Directive proposal, Article 81(2)). In its explanatory memorandum, the Commission has considered it important to specify that this baseline protection "remains competitive given what other regions offer".
Three options were initially considered including the possibility to maintain the current system and add conditional protection periods. The option related to variable duration of regulatory data protection (split into baseline of 6 years and additional periods) was however preferred and considered as "a cost-effective way of achieving the objectives of improved access". Pharma companies have already expressed its concern arguing that this period is essential to stimulate the development of innovative therapies.
The period of market exclusivity (e.g. the period during which a generic/biosimilar/hybrid of the initially authorised product cannot be placed on the market) remains for a period of two years after the expiry of the relevant regulatory data protection periods2.
Lastly, it should be noted that the data and market protections may be suspended in the case where a compulsory licence granted by a relevant authority in the Union to a party, to address a public health emergency during the period of such licence.
1.2 Additional data protection period
While the Commission proposes a reduction of the baseline period of data protection period, the proposed legislation also diversifies the possibilities for additional prolongation of this period of protection.
Medicinal products supplied in Member States. The data protection period could be prolonged by 24 months (Directive proposal, Article 81,2 a), if, within 2 years3 from the date of granting of the MA, the MA holder fulfils the following criteria:
The supply of the medicinal product. Medicinal products should be released and continuously supplied into the supply chain in a sufficient quantity and in the presentations necessary to cover the needs of the patients medicinal products in the Member States in which it is valid.
The type of MA. Medicinal products should be authorised via a centralised or decentralised procedure.
The variation application (Directive proposal, Article 82). To obtain such a prolongation, the MA holder should apply for a variation of the MA. Such variation application, which, in principle, would have to be submitted between 34 and 36 months after the date of granting of the MA4, would have to include documents from Member States in which the MA is valid either confirming the released and continuously supplied of the product in its territory or waiving the condition of launch in their territory for the purpose of the prolongation5.
This procedure would necessarily lead to an additional workload for pharmaceutical companies as they will have to request the appropriate documentation to the Member States which will then have 60 days to respond. In case a Member State does not reply this would be considered as the statement of non-objection has been provided.
Implementing measures related to those procedural aspects and to the criteria of released and continuously supply may be adopted by the Commission. Regarding, in particular, the term "continuously supplied", clarification will be needed, notably in connection with the new obligations intended to prevent shortage.
In any case, this condition for prolongation of data protection seems difficult to meet and will necessarily imply an early strategic reflection concerning the countries in which the MA will have to be submitted in the case of a decentralised MA6. As far as a centralised MA will be concerned, the burden for the MA holder to market the product, in all Member States, will certainly imply substantial challenges and could even lead to supply issues.
Unmet medical need. The data protection period could be prolonged by 6 months if the MA holder demonstrated that the medicinal product addresses unmet medical needs7 (Directive proposal, Articles 81,2 b and 83), knowing that designated orphan medicinal products shall be considered as addressing an unmet medical need.
The product must then fulfil the following conditions (Directive proposal, Article 83):
At least one of the indications relates to a life threatening or severely debilitating disease.
No medicinal product is authorised for this disease in the Union or the disease is associated with a remaining high morbidity or mortality despite the presence of existing authorised products.
The administration of the concerned medicinal product results in a meaningful reduction in disease morbidity or mortality for the relevant patient population.
The EMA should adopt scientific guidelines for the application of this criterion concerning unmet medical need which are absolutely necessary considering the imprecision of the provisions. In particular, the notion of meaningful reduction is relative and is likely to be interpreted in different way depending on the pathologies.
New active substance. A prolongation of 6 months of the data protection period would be granted for medicinal products containing a new active substance, provided that the clinical trials supporting the initial MA application use a relevant and evidence-based comparator. A scientific advice will be provided by the EMA (Directive proposal, Article 81,2 c).
Additional therapeutic indication. The data protection period may be extended by 12 months in the case the MA holder obtains, during the data protection period, an authorisation for an additional indication for which it has demonstrated a significant clinical benefit in comparison with existing therapies. As a reminder, a similar prolongation already exists in the current legislation (Directive proposal, Article 81,2 d).
2. Particular situations concerning specific products
Beyond the data protection and market exclusivity granted to medicinal products as described above, some medicinal products are, in the recent legislative proposals, subject to specific provisions.
Repurposed products (Directive proposal, Article 84). A data protection period of 4 years "shall be granted for a medicinal product with respect to a new therapeutic indication not previously authorised in the Union, provided that":
A significant clinical benefit has been demonstrated through adequate non-clinical or clinical studies conducted in relation to the therapeutic indication; and
The medicinal product has been authorised through an abridged application (Directive proposal, Articles 9 to 12) and has not previously benefitted from data protection, or 25 years have passed since the initial MA granting.
Such protection period would only be granted once for a specific medicinal product. The MA will have to mention, during such data protection period, that the medicinal product already exists and has been authorized with an additional indication.
Priority antimicrobials (Regulation proposal, Articles 40 et 41). One of the objective of the pharmaceutical package is to enhance the development of priority antimicrobials. Therefore, antimicrobials are subject to numerous measures, including concerning data protection.
An applicant for a MA concerning a priority antimicrobial may request a transferable data exclusivity voucher, which granting is submitted to strict criteria, which would grant an additional 12 months of regulatory data protection.
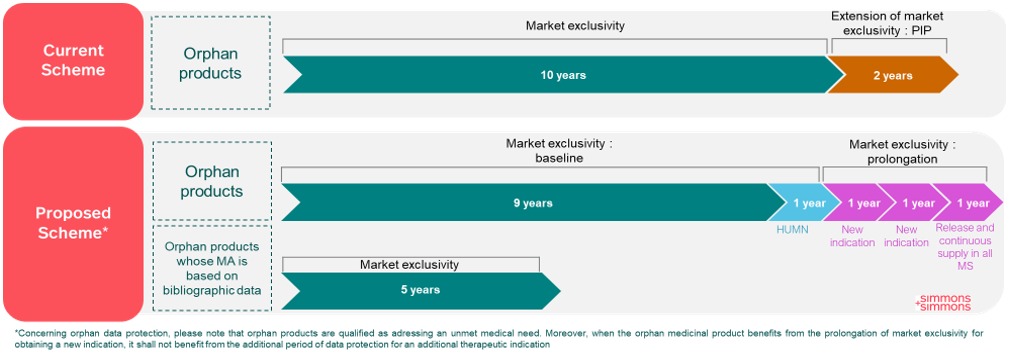
Orphan medicinal products (Regulation proposal, Articles 71 and 72).
The duration of market exclusivity for orphan products is, as follows:
10 years for orphan medicinal products addressing a high unmet medical need;
5 years for orphan medicinal products whose MA is based on bibliographic data; and
9 years for others orphan medicinal products.
Such market exclusivity shall start from the date when the first orphan MA, for a same active substance, was granted in the Union.
The market exclusivity period can be prolonged for orphan products with a MA not based on bibliographic data:
For an additional 12 month exclusivity, if the orphan product is released and continuously supplied (and fulfils the conditions of Directive proposal, articles 81(2)a and 82(1), see above)
For an additional 12 months exclusivity, if the MA holder of the orphan product obtains a MA for one or more new therapeutic indications (for a different orphan condition) at least two years before the end of the exclusivity period. This prolongation may be granted twice.
When the orphan medicinal product benefits from such prolongation of market exclusivity, it shall not benefit from the additional period of data protection for an additional therapeutic indication for which the MA holder has demonstrated, with supporting data, a significant clinical benefit in comparison with existing therapies (referred to in Article 81(2), point (d) of Directive proposal).
Paediatric-use marketing authorisation ("PUMA"). As a reminder, a PUMA is a MA developed for use in paediatric population and concerns medicinal products already authorised and no longer covered by a supplementary protection certificate.
According to the Regulation proposal, a medicinal product benefiting from a PUMA including the result of all studies complying with the agreed paediatric investigation plan may benefit from independent data and marketing protection periods (Regulation proposal, Article 93).
Our next article will focus on the Bolar exemption and the requirements for generics and biosimilars.
1 The Directive proposal includes transitional provisions (Article 218.5): reference medicinal products for which an MA application has been submitted before a delay 18 months after the date of entry into force of Directive proposal, shall be subject to the provisions on data protection periods laid down in Article 10 of Directive 2001/83/EC
2 Please note that market protection shall be distinguished from market exclusivity :
Market protection corresponds to the period during which, applications for generics/biosimilars/etc. can be accepted and authorised, but cannot be placed on the market. The starting point of this protection is the expiry of the relevant regulatory data protection periods.
Market exclusivity corresponds to the period during which, generics/biosimilars/etc. of an orphan product cannot be placed on the market. The starting point of this protection is the date when the first orphan MA, for a same active substance, was granted in the Union
3 The period to fulfil this criteria is extended to 3 years for some entities:
SMEs;
Not-for-profit entities; and
Entities, which, at the time of the granting of the MA, have not receive more than 5 centralised MA or whose group has not received more than 5 centralised MA.
4 This delay if extended to 46-48 months for SMEs, not-for-profit entities and entities, which, at the time of the granting of the MA, have not receive more than 5 centralised MA or whose group has not received more than 5 MA.
5 The explanatory memorandum of the Directive proposal specifies that the Member State may waive the launch of the product in its territory when the launch is materially impossible or if the Member State wishes the launch to intervene at a later date. Moreover, the recital 57 of the proposal Directive details that the waiver does not affect the powers of the Member States concerning the supply, fixing the prices or the reimbursement of the concerned medicinal products nor does it imply that the Member State has waived its possibility to request release or supply of the product concerned at any time before, during or after the prolongation of the data protection period.
6 Please note that according Article 34 (3) of the Directive proposal, "the competent authority of a Member State may request for justified public health reasons to enter the procedure and shall inform the applicant and the competent authority of the reference Member State for the decentralised procedure of its request within 30 days from the date of submission of the application".
7 Please note that for conditional MA, the prolongation of the data protection period regarding unmet medical needs would only apply if within 4 years of the granting of the conditional MA, the medicinal product has been granted a full MA.




.jpg?crop=300,495&format=webply&auto=webp)








.jpg?crop=300,495&format=webply&auto=webp)

.jpg?crop=300,495&format=webply&auto=webp)




