In what seems to be becoming the most controversial aspect of recent EU SPC reform proposals, the new centralised examination procedure and unitary SPC title ("uSPC") are accompanied by a number of new ways to challenge SPC validity. These include: (i) formal pre-grant third party observations; (ii) a new pre-grant SPC opposition procedure; and (iii) a new form of post-grant SPC invalidity proceedings. We review the proposed procedures and consider the implications for parties who typically hold and/or challenge SPCs.
Is this all familiar ground?
These procedures are familiar ground for SPC practitioners given the close connection patent law and practice, which brings parallels to centralised examination before the European Patent Office (EPO).
Moreover, third party observations (3POs) already form part of established SPC examination practice before a number of national authorities and provide a relatively straightforward approach to file validity objections to try to prevent or delay grant, although without bringing the third party filer into proceedings. Accordingly, 3POs seem to be the less controversial aspect of the reform proposals, and we do cover them in more detail here (and we have mentioned them in our previous article about examination - see here). Instead we focus on the two more controversial proposals, namely:
First, while are also familiar from the EPO (at least post-grant), both the introduction of oppositions into SPC procedure and their timing (pre-grant) have courted controversy.
Second, is the introduction of a post-grant invalidity procedure. While limited to the uSPC, having a centralised validity option is familiar only perhaps to brands practitioners and current EU IPO users, rather than patent/SPC practitioners.
Overall, the prospects of pre-grant objections, potential delays to grant and a new authority governing SPC validity have raised concerns with SPC applicants versus a welcome from SPC challengers.
The battle lines over oppositions were predictable
Centralised examination of EU SPCs received almost unanimous support, including both sides of the usual pharma divide between innovators (i.e. typical SPC applicants) and generics/biosimilars (i.e. typical SPC challengers).
Centralised examination brings with it an inevitable consideration of centralised opposition, on which the usual pharma divide returns. This was illustrated in the feedback during the early stages of the EU work programme, where, for example, EFPIA strongly opposed oppositions compared to Medicines For Europe strongly advocating their benefits.
Central to this debate is another new development, and indeed partial driver for these very SPC reforms, namely the advent of the Unified Patent Court (UPC) in Europe (open since 1 June 2023). In this way, there is an argument that the UPC would be the natural forum for addressing harmonised cross-border disputes concerning SPC validity (including uSPCs provided there is an amendment to the UPC Agreement, which the reform proposals mention would be necessary). Jurisdiction of the UPC over SPCs was already contemplated in association with Unified Patents (UPs) and non-opted out European Patents (EPs). The significance of SPCs at the UPC was further endorsed as they were itemised as an apparently independent jurisdiction to be allocated to Paris (i.e. as part of the reallocation of London's responsibilities to the forthcoming trifecta of central divisions in Paris/Munich/Milan).
The arising tension has therefore been whether the new central SPC opposition unnecessarily erodes what was otherwise expected (or hoped) would become part of the UPC's (eventually exclusive) jurisdiction. However, in the explanatory memorandum to the new Regulations, the reason for the opposition procedure is said to be to avoid national litigation. This would suggest it was merely to unify litigation, with the balance arguably in favour of SPC challengers (compared to SPC applicants, who take all the validity risk).
Other reasons for proposing a central opposition procedure may also have been to provide a forum with precisely commensurate jurisdiction (as UPC contracting states are only a portion of EU member states). Furthermore, another utility of centralised SPC opposition is the ability for anyone to file an opposition, including any EU member states. This means that the SPC opposition procedure arguably creates a fully harmonised system in which input from all member states may be taken into account (replacing the current national influence via national filing practices and the manner in which EU Member States may otherwise submit views primarily as part of any CJEU referral procedure).
Pre-grant oppositions - how they work
A basic outline of the SPC opposition procedure is set out below.
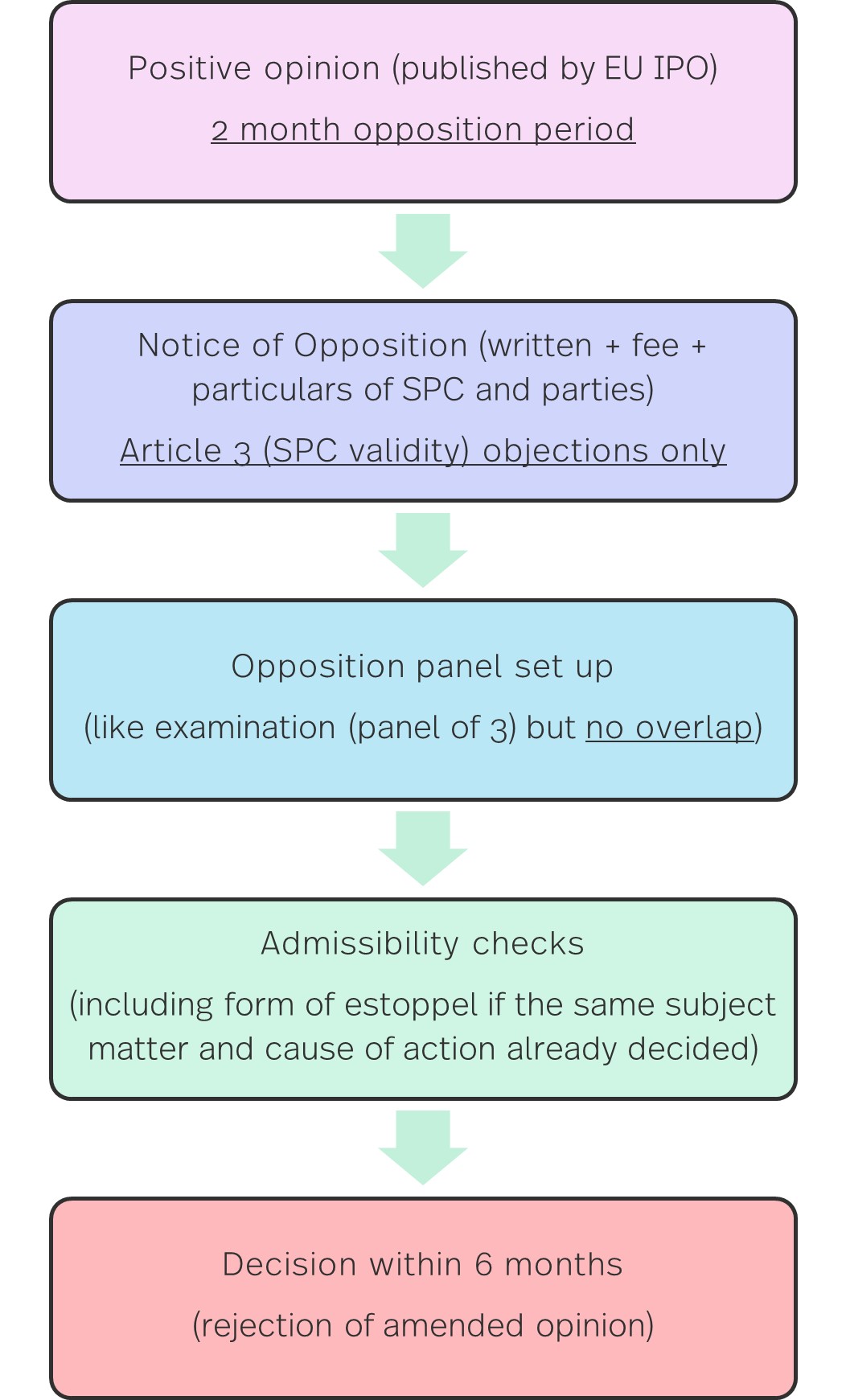
A key message is the onus on speed with decisions said to be given within 6 months. However, this is caveated if the proceedings are "complex", which is something that could arguably be said of many SPC disputes. Indeed, the new proposals include detailed provisions for admitting evidence, expert opinions (including hearing experts), etc. So it remains to be seen whether this timeframe is realistic. Plus of course there is the likelihood of an appeal. While SPCs tend to be applied for years before they enter into force, concerns over the timeframe of this procedure are likely to remain.
Otherwise, there are some key questions: for example, will 'strawmen' be able to file SPC oppositions (like the EPO practice)? And will oppositions about SPCs risk adverse implications for patent validity, which again may not be raised in litigation until a number of years later? While oppositions are restricted to SPC validity, including the key 4 pillars under Article 3(a)-(d), there are inevitably questions that might overlap with patent validity, such as the independent inventiveness of combinations. But if impact on patent validity is a concern in SPC oppositions, this is amplified under the new invalidity procedure for uSPCs, which expressly introduces it. We look at this next.
Post-grant validity of uSPCs
If proposals for pre-grant SPC opposition are perceived as nibbling at the UPC's contemplated SPC (and uSPC) jurisdiction, then the proposal for a central invalidity challenges to uSPCs takes a proper, bigger bite.
The procedure is similar to oppositions, including with respect to submissions and expert evidence, the time constraints of a decision within 6 months (unless complex) and the ability to appeal. But there are also a few particular points that jumped out as significant.
i. Patent validity - First, if the underlying patent has expired, the proceedings can assess the validity of the patent underlying the uSPC (i.e. on the principle that if patent is invalid, so is the uSPC). While the timing in itself appears to seek to avoid a clash with patent litigation, this is not necessarily the case (as patent expiry does not always mark the end of litigation). So this could lead to central assessment of patent validity either: in parallel with national patent litigation proceedings (if ongoing); as a re-assessment (if national litigation has concluded); or as de novo proceedings (if national litigation has never occurred); and all in a completely new forum. As well as the timing and justification for this procedure, concerns seem almost certain to be raised over the suitability of allocating this jurisdiction to EU IPO, which is already new to SPCs. Ultimately, given this issue is specific to uSPCs, it could also risk a significant lack of uptake of what was otherwise hoped would be a key unitary right to accompany the unitary patent.
ii. Multiple/parallel proceedings - Second, aside from the issue of patent validity, the proposals include mechanisms for dealing with multiple or parallel proceedings concerning SPC validity between the same parties. In this way, there are reflective proposals which either prevent the EU IPO proceeding with a uSPC invalidity claim (e.g. if the same parties have litigated the same subject matter and cause of action in national litigation in a Member State to a final decision) or the converse (i.e. preventing Member States from proceeding with a counterclaim if it has already been decided by EU IPO). These proposals seem to be an attempt to create a form of binding precedent and/or estoppel between the same parties and so present interesting jurisdictional questions. As well as preventing parallel or multiple proceeding across the EU, this may, for example, be intended to stop parties from re-litigating in the EU IPO (e.g. if an opposition is defeated, the same party would be stopped from seeking a declaration of invalidity at the EU IPO post grant).
iii. Other regulatory rights - Third, while venturing into patent validity already seems likely to be controversial, the proposals - again only in respect of uSPCs - also allocate jurisdiction to the EU IPO to consider the validity of 6 month 'paediatric' extensions to SPC term granted under Regulation 1901/2006. This would introduce another area of jurisdiction, albeit where litigation is less common.
Final comments
There is plenty of detail to get into in these EU SPC reforms but the validity procedures draw a significant amount of attention, as can already been seen in the feedback to the proposed regulations (for which the deadline is 21 September). Assuming a drastic U-turn is unlikely, SPC applicants and challengers will otherwise have to plan for the creation of a significant new forum for determining SPC validity, which might even risk spilling over to patent validity (and paediatric extensions) for uSPCs. Time will tell whether this new system might eventually be embraced (or at least considered unavoidable), or whether applicants will devise strategies seeking to avoid central SPC examination and/or uSPCs altogether in order to escape any concerns and uncertainties.




.jpg?crop=300,495&format=webply&auto=webp)







_11zon_11zon.jpg?crop=300,495&format=webply&auto=webp)


_11zon.jpg?crop=300,495&format=webply&auto=webp)




.jpg?crop=300,495&format=webply&auto=webp)