Part 1 - T 178/23 - Relevance of the Decision and General Principles
T 178/23 underlines the general principle at the EPO that a reinstatement or re-establishment of rights is only available if there was an intention to meet a respective deadline, but the deadline was missed due to an isolated error. A reinstatement is not available in cases where there was no intention to meet the deadline (in the present case, the filing of an appeal), irrespective of whether this was due to an error or not.
Furthermore, T 178/23 underlines again that it is very important to review thoroughly the documents accompanying the Communication pursuant to R. 71 (3) EPC, since it is very difficult to make any further changes after filing the approval to the documents intended for grant.
Lastly, T 178/23 summarizes the requirements for a request for a reinstatement and so is worth reading also for this reason. Especially, the decision makes clear that all arguments in support of a reinstatement need to be filed with the request for reinstatement or at least within the respective deadline for such a request.
The reasons/decision
In the case underlying T 178/23, the drawings were missing in the documents intended for grant accompanying the Communication pursuant to R. 71 (3) EPC. The Applicant did not notice this and approved the text. Only after expiry of the deadline for an appeal against the decision to grant, the Applicant filed an appeal against this decision. A request for reinstatement into the term for filing an appeal and the payment of the appeal fee was then filed later, after the Board had notified the Applicant that the deadline for filing an appeal had been missed. From the request of reinstatement and Applicant's further submissions in the appeal procedure, it was clear that Applicant had not noticed that the drawings were missing, but had only been informed on this issue upon validation proceedings in Italy.
The Board rejected the Applicant's request for a reinstatement, because it found that the request had not been substantiated. Furthermore, the request was also rejected because the reason for not filing the appeal was not an individual error which occurred despite of the fact that Applicant has taken all necessary steps to ensure that the deadline was met. Rather, the reason was that the Applicant did not appreciate that the drawings were missing. So, in fact, Applicant did not intend to file a notice of appeal, and a reinstatement is not available in such cases. This already follows from the wording of Art. 122 (1) EPC and has been confirmed in various TBA decisions.
The circumstances of the present case are especially unfortunate for the Applicant since the drawings were missing in the documents intended for grant due to an error of the European Patent Office. Despite efforts taken by the office, it still happens too often that the documents intended for grant contain errors, especially in cases where the application is adapted to the allowable claims by the Examiner. If such errors are not spotted by the Applicant, they can only be corrected after the expiry of the term for filing an appeal against the decision to grant in case of typographical errors. T 178/23 highlights this unfortunate situation, and points to the fact that a careful review of the documents intended for grant is absolutely essential.
Take Home Message
A reinstatement or re-establishment of rights is only available in cases where there was an intention to meet a deadline before the EPO, but the deadline was not met in spite of all due care taken by the Applicant or Patentee. Furthermore, a careful review of the documents intended for grant is absolutely essential, since it is very difficult to correct these documents later.
Part 2 - UPC 'bifurcation' trend continues
In our article published in JUVE about 1 year ago (here), we discussed a bifurcation trend emerging from defendant-driven strategies in some of the big pharma/biotech and medtech cases at the UPC; in particular arising from an apparent desire to steer validity proceedings to the Central Division (CD) and away from Local Divisions (LDs).
The two early cases we discussed (Amgen v Sanofi and Meril v Edwards) involved the then two CDs in Munich and Paris. Not long after its establishment, activity at the third CD in Milan continues to evidence this trend. And a recent decision of the Milan CD addressed a key issue about timing actions in parallel divisions. We discuss these developments below.
Revocation actions in Milan
Since June 2024, the Milan CD was established and took jurisdiction, inter alia, over UPC revocation actions for patents (without supplementary protection certificates) classified under human necessities, comprising in particular pharmaceutical and medical devices and also agriculture, food, tobacco, etc.
According to a recent search of the UPC's CMS, there are 6 visible revocation actions in Milan (i.e. those where the actions are understood to have been served, and more could be issued/pending):
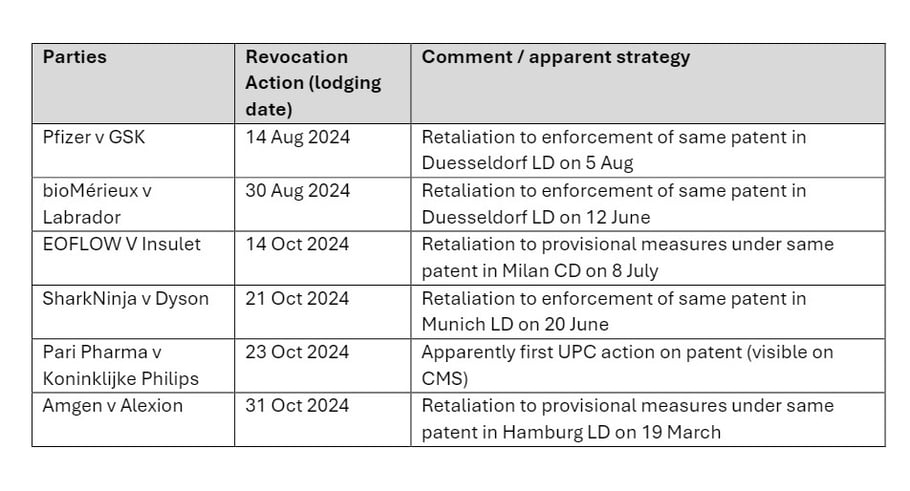
While this is a small data set, these actions continue to display a trend of 'bifurcation' by defendants commencing parallel proceedings in the Milan CD, notwithstanding the patentee's pursuit of infringement actions via various LDs. They also continue to display this trend in pharma/biotech and medtech cases, as well as other fields, such as consumer products.
In particular, all but 1 of the revocation actions are seemingly retaliatory; 3 out of 6 follow LD enforcement actions and 2 out of 6 follow LD actions for provisional measures. In only 1 of the actions is the revocation action apparently the first UPC action on the patent in question.
Focussing on the 3 revocation actions following LD enforcement actions, the gap is quite variable - one followed within days (Pfizer v GSK - discussed below regarding the recent decision in this case), whereas the other two had a gap of a few months. We discuss the 2 cases with a longer gap first, before moving to the Pfizer case.
Retaliating within a few months
The frontrunner case of Meril v Edwards that we discussed previously is a closer parallel to the latter two cases as the gap is longer; in Meril, the CD revocation (then in Paris) was commenced in August 2023, approx. 2 months after the Munich LD enforcement action (lodged on UPC opening day, 1 June 2023).
Recapping on what Meril achieved and so illustrating the potential road ahead and corresponding objectives of these Milan CD revocation actions, the following strategic issues are of note:
1) A validity-only trial 10m from claim - Meril's revocation action (including a rejection of Edwards' preliminary objection to this strategy) proceeded quickly, and after March 2024, included the counterclaims for revocation from the Munich LD action (which were referred to the Paris CD under Article 33(3)(b) UPCA). The consolidated trial was heard in the Paris CD on 7 June 2024.
2) A CD only trial - The ensuing decision of the Paris CD (of 19 July 2024) refers to the consolidation and disagrees with the Munich LD as to its suggestion that Rule 340 RoP (about 'connection joinder') could be interpreted to require a joint hearing for the revocation proceedings before a panel comprising all judges from both the central and local divisions. In the Paris CD's view, referral under Article 33(3)(b) does not result in a "true merger" of the cases, which therefore "retain their distinct legal identities and must be adjudicated independently, even if a single decision is ultimately issued".
3) A shot at invalidity months in advance of an infringement trial - when the Paris CD's decision of 19 July 2024 from the 4 June hearing was delivered, the Munich LD infringement action was ongoing, albeit slower. The LD action then had to take into account the Paris CD decision (upholding Edwards' patent in amended form) as well as the revocation of a related patent at the European Patent Office. The LD infringement trial eventually took place on 24 September 2024 (3 months after validity), and in its decision dated 15 November 2024, Meril were held to infringe and so were injuncted (subject to a partial disclaimer).
While unsuccessful to date, the third point is the most significant as Meril's strategy ultimately got them a shot at invalidity before infringement was decided. It now remains to be seen if the claimants in the more recent Milan CD actions will achieve the same speed, timing and opportunity so to land their invalidity attacks first, and indeed whether this trend will continue to appear at the UPC.
Notably in both of the recent Milan CD actions, the 'defendants' in the enforcement proceedings have commenced revocation actions via group entities that were not sued in the LD infringement proceedings (SharkNinja via an Italian entity, and bioMérieux via a UK entity). Both cases appear to be proceeding with, so far, no record of a preliminary objection on the CMS. The third case, mentioned above and involving Pfizer and GSK is more interesting in this respect as there are overlapping parties in the actions. We address this case next, including the issue of timing.
The significance of timing and the perils of a false start?
Moving to the 3rd of the 3 recent revocation actions at the Milan CD which appear to be 'retaliatory' to local enforcement proceedings, Pfizer commenced its revocation action only days after being sued by GSK in the Düsseldorf LD. A recent decision of the Milan CD in this case, dated 2 December 2024, reveals the significance of this timeline, which is briefly summarised as follows:
- GSK sued Pfizer for infringement in Düsseldorf LD on 5 August 2024, after the issuance of the EPO's intention to grant its patent; but before publication of grant (on 14 Aug 2024) and before the date of registration of the patent's unitary effect (on 22 Aug 2024, retrospectively applying to 14 Aug 2024);
- Pfizer sued to revoke GSK's patent in Milan CD on 14 Aug 2024, 9 seconds after midnight on the date of publication of grant;
- Both parties filed preliminary objections to the other's actions (GSK also claiming that Pfizer's preliminary objection was too late and inadmissible).
In essence, this scenario seems to be a close parallel to the second of the earlier cases we reviewed, namely Amgen v Sanofi. We discussed that case as a race to file on Day 1 of the UPC, which Sanofi won and accordingly first seized the Munich CD. In terms of what this strategy achieved in that case, Sanofi went on to consolidate its 'validity only' trial at the Munich CD (alongside Regeneron's counterclaim from the LD action) and - like Meril - they ultimately got their shot at invalidity months in advance of the infringement trial. But unlike Meril, Sanofi/Regeneron succeeded in revoking Amgen's patent and so the infringement action came to be stayed.
In Pfizer v GSK, it appears that Pfizer was ready for the same kind of race on 14 August (the date of publication of grant), which (with the CMS operating) it completed in only 9 seconds. In doing so, Pfizer claimed that GSK's completion over a week earlier was a false start, thereby (to continue that metaphor) claiming victory by way of disqualification.
In addition to the comparison to Amgen v Sanofi and as mentioned above, there are also interesting issues relevant to the Meril v Edwards case, as, unlike Meril, Pfizer issued its revocation action in the name of claimants that had also been sued in GSK's enforcement action (whereas Meril's claimant for its revocation was an Italian company that had not been sued by Edwards, as now repeated in principle by SharkNinja and bioMérieux). The Milan CD's decision addresses the overlap of the parties first, before turning to the timing/false start issue. We summarise the findings on the two central issues as follows:
Overlapping/new parties - GSK had sued 14 Pfizer EU entities in its LD action, which all appeared to have been served with GSK's infringement proceedings in advance of Pfizer's revocation action. Pfizer's revocation action was lodged by 6 of those 14 entities, along with 2 new entities: Pfizer Inc (a US company) and Pfizer Ltd (a UK entity). The Milan CD, referring to the Paris CD's decision in Meril v Edwards (similarly dismissing Edwards' preliminary objection) held that the 2 new Pfizer parties are not the "same parties" and so could proceed with their revocation action in any event.
Was GSK's infringement action too early (or a false start)? - Of greater significance perhaps is the Milan CD's view that the UPC did not have jurisdiction over GSK's patent when it commenced its infringement action as it was before grant. The decision records GSK's objections in this respect, including that other UPC actions (e.g. by 10x and Alexion) had been commenced before grant, that grant was 'set in stone' by the date of GSK's action (as it was after intention to grant the patent ) and that Pfizer had commenced its action in full knowledge of GSK's infringement action to circumvent and frustrate the otherwise clear provisions, including by using claimants outside UPC/EU member states who were therefore not 'concerned' with the enforced patents. However, the Milan CD dismissed all of these arguments and in doing so dismissed GSK's preliminary objections, thereby finding that it could proceed with the revocation action by all of the Pfizer claimants.
So what remains interesting in these parallel UPC proceedings is what the Düsseldorf LD will have to say, as it's view on Pfizer's preliminary objection is presumably pending (despite being filed 1 month earlier than GSK's objection in the Milan CD). In this respect, of most interest will be the Düsseldorf LD's view on whether GSK's action was inadmissible (as the Milan CD concludes) and also whether it could 'mature' into an admissible action following patent grant.
In any event, the Milan CD expressly concluded that it does not need to wait for the Düsseldorf LD's decision as it viewed the initial inadmissibility as fatal to GSK's objection, inferring that both actions could still proceed in parallel. The Milan CD also addressed another issue of overlap, as one of the other grounds relied upon by GSK was that Pfizer had submitted to the jurisdiction of the Düsseldorf LD by not filing a preliminary objection on time, on which the Milan CD agreed with Pfizer's position that its objection was on time; in any event, the Milan CD concluded this was insufficient evidence of submission to jurisdiction so that it would not matter if the Düsseldorf LD took a different view.
The Milan CD decision also covers a number of significant issues, including that this decision could be decided without a hearing, which may have contributed to the speed of this decision (including potentially that it came out before the Düsseldorf LD's decision on its preliminary objection). The Milan CD also refused all of GSK's requests to stay (either pending the preliminary objection in Düsseldorf or the Düsseldorf action itself).
Overall, this case seems very likely to be heading to the UPC Court of Appeal for further guidance on the use of this strategy by Pfizer, making this a key case to watch as this 'bifurcation' trend continues to emerge at the UPC.







_11zon_11zon.jpg?crop=300,495&format=webply&auto=webp)


_11zon.jpg?crop=300,495&format=webply&auto=webp)


.jpg?crop=300,495&format=webply&auto=webp)


.jpg?crop=300,495&format=webply&auto=webp)

.jpg?crop=300,495&format=webply&auto=webp)