The Dutch Central Transparency Register in a nutshell
The Dutch authorities are pushing for transparency between the industry and healthcare providers.
Cooperation between the industry and prescribers is widely considered to be necessary in order to advance the development of medicines, medical devices and safe and efficient use. This contributes to the improvement of healthcare in general. However, secrecy about the existence of financial relationships undermines patient confidence in healthcare and in the integrity of physicians. It is therefore useful for parties to be open about this. For this reason, all stakeholders involved in the provision and delivery of healthcare in The Netherlands agreed to lift the secrecy surrounding cooperation between healthcare providers and the industry.
The Dutch Central Transparency Register
Since 25 April 2013 financial interactions between individual Health Care Providers and Organisations (HCP’s and HCO’s) must be disclosed in the online Dutch Central Transparency Register (DCTR).
Initially, the DCTR was set up to disclose interactions between the pharmaceutical industry and HCP’s but as of 2016 the regime of transparency has started to apply to the medical devices sector as well, covering the full sector as of 2017.
The DCTR is governed and operated by the Foundation Transparency Register Care (Stichting Transparantieregister Zorg).
Legal background
Typically (and perhaps strangely), the DCTR has no formal basis in Dutch legislation, it is based on participation of stakeholders in the medical devices and pharmaceutical industry to two Industry Codes: the CGR Code (Pharmaceutical Advertising Code) and the GMH (Code of Conduct Medical Devices). Through memberships, these codes cover the Dutch pharmaceutical and medical devices industry as well as HCP’s registered in The Netherlands by and large.
The Transparency Register Foundation and both Industry Codes have advocated the well functioning of self-regulation of the LS sector in The Netherlands, which is indeed well embedded and highly esteemed in The Netherlands. The Ministry of Health has publicly endorsed the DCTR but is closely monitoring that the DCTR de facto leads to the disclosures as intended. Up until today, the Ministry has seen no reason to interact which implies that the system functions well.
The key elements of the DCTR will be outlined below.
Financial relationships
A financial relationship is defined as any direct or indirect financial or other compensation measurable in terms of money, made to healthcare providers residing or practicing in the Netherlands, either sole practitioners, associations or healthcare institutes.
All financial relationships between the pharmaceutical and medical devices industry and healthcare providers must be recorded in written agreements. Those written agreements do not need to be disclosed, only the financial agreements and type of services provided.
General purpose of the DCTR
The purpose of the central register is to enable consumers to obtain up-to-date information on existing financial links. This information can be relevant in discussions between patients and doctors and decisions about the use of medicines.
As a result, the DCTR is searchable on a HCP’s name and BIG registry number only.
Obligation to disclose
The written agreement must specify which party will bear the responsibility to adhere to the transparency obligations. Still, in case of a relationship between an HCP and foreign company who may not be a member of the Dutch Industry Codes, the obligation to disclose would rest with the HCP.
The following information must be provided:
- the nature of an agreement following a standardized format;
- name and place of residence/registration number chamber of commerce of the marketing authorization holder;
- effective period of the agreement; and
- the total amount of fee payments made to an individual healthcare practitioner or the total amount of payments made to the association or institute whit whom financial relationships exist.
Data retention and privacy
An important aspect of the Code of Conduct is that the data disclosed will be removed from the register three years after services have been terminated. Although this seems to be detrimental to the objective of transparency, this term was considered to strike a balance between legitimate privacy protection concerns of doctors who’s names are listed whilst maintaining an adequate level of up-to-date transparency at the same time.
Trends in financial reportings
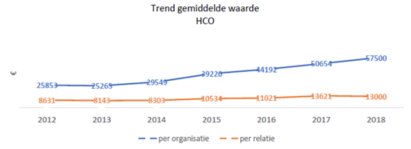
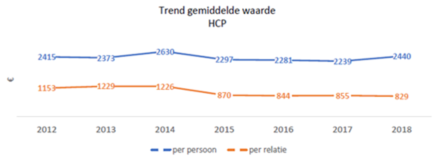
The figures show that the size of financial relationships with individual care providers is fairly stable: an average of approximately €2,200 per year. However, healthcare institutions do show an increase in the average amount received. This growth is mainly visible at academic hospitals, and is a direct result of the participation of all suppliers of medical devices since 2017.
Average value of receivables HCP’s (2012 – 2018, as disclosed by the Foundation (Stichting Transparantieregister Zorg))
Average value of receivables Health Care Institutes (2012 – 2018, as disclosed by the Foundation (Stichting Transparantieregister Zorg))